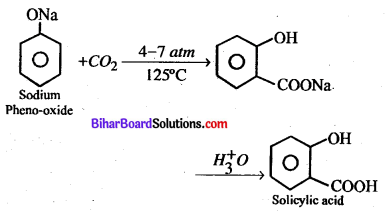
Bihar Board 12th Biology Objective Questions and Answers
Bihar Board 12th Biology Objective Answers Chapter 4 Reproductive Health
Question 1.
RCH stands for
(a) routine check-up of health
(b) reproduction cum hygiene
(c) reversible contraceptive hazards
(d) reproductive and child health care.
Answer:
(d) reproductive and child health care.
Question 2.
Amminocentesis is a technique used to
(a) determine errors in amino acid metabolism in embryo
(b) pin point specific cardiac ailments in embryo
(c) determine any hereditary genetic abnormality in embryo
(d) all of these.
Answer:
(a) determine errors in amino acid metabolism in embryo
Question 3.
Which of the following cannot be detected in a developing foetus by amniocentesis ?
(a) Jaundice
(b) Down’s syndrome
(c) Cystic fibrosis
(d) Colourblindness
Answer:
(a) Jaundice
![]()
Question 4.
In which of the following weeks of pregnancy CVS is done ?
(a) 12th – 14th week
(b) 8th – 10th week
(c) 5th – 7th week
(d) None of these
Answer:
(b) 8th – 10th week
Question 5.
Causes for increased population growth in india is/are
(a) increase in birth rate
(b) decrease in death rate
(c) lack of education
(d) all of these.
Answer:
(d) all of these.
Question 6.
Which of the following pairs contributes to an increase in population ?
(a) Natality and immigration
(b) Mortality and emigration
(c) Natality and emigration
(d) Mortality and immigration
Answer:
(a) Natality and immigration
Question 7.
Which of the following is correct regarding the consequences of lower population ?
(a) It increases the poverty of a country.
(b) It leads to shortage of food supply.
(c) It results in unemployment
(d) All of these
Answer:
(d) All of these
Question 8.
The best way to decrease population of a country is
(a) to educate people
(b) to have better houses
(c) to kill people on a large scale
(d) to practice and implement familay planning.
Answer:
(d) to practice and implement familay planning.
Question 9.
Which of the following correctly describes the measures
that can be used to control over-population ?
(a) Educating people about the advantages of a small family
(b) Raising the age of marriage
(c) Encouraging family planning programme
(d) All of these
Answer:
(d) All of these
![]()
Question 10.
Which of the following is not a characteristic of an ideal contraceptive ?
(a) User-friendly
(b) Irreversible
(c) Easily available
(d) Least side-effects
Answer:
(b) Irreversible
Question 11.
On which of the following facts does the method of periodic abstinence is based ?
(a) Ovulation occurs on about the 14th day of menstruation.
(b) Ovum remains alive for about 1-2 days.
(c) Sperms survive for about 3 days.
(d) All of these
Answer:
(d) All of these
Question 12.
Which of the following is a non-medicated intrauterine device (IUD) ?
(a) CuT
(b) Lippes Loop
(c) Cu7
(d) LNG-20
Answer:
(b) Lippes Loop
Question 13.
Multiload device contains
(a) manganese
(b) iron
(c) copper
(d) calcium
Answer:
(c) copper
![]()
Question 14.
Which of the following is not an intrauterine device ?
(a) Progestasert
(b) Multiload-375
(c) Norplant
(d) Lippes loop
Answer:
(c) Norplant
Question 15.
What is the work of copper-T ?
(a) To inhibit ovulation
(b) To inhibit fertilisation
(c) To inhibit implantation of blastocyst
(d) To inhibit gametogenesis
Answer:
(b) To inhibit fertilisation
Question 16.
IUDs prevent pregnancy by
(a) inhibiting physiological and morphological uterine changes required for implantation.
(b) increasing phagocytosis of spermatozoa within uterus
(c) suppressing motility of sperms as well as their fertilising capacity
(d) all of these.
Answer:
(d) all of these.
Question 17.
Cu ions released from copper releasing intrauterine devices (IUDs)
(a) prevent ovulation
(b) make uterus unsuitable for implantation
(c) decrease phagocytosis of sperms
(d) suppress sperm motility.
Answer:
(d) suppress sperm motility.
Question 18.
The birth control device used by women is
(a) diaphragm
(b) vault
(c) copper T
(d) all of these.
Answer:
(d) all of these.
![]()
Question 19.
The most important component of oral contraceptive pills is
(a) progesterone-estrogen
(b) growth hormore
(c) thyroxine
(d) luteinising hormone.
Answer:
(a) progesterone-estrogen
Question 20.
Progesterone pill helps in preventing pregnancy by not allowing
(a) ova formation
(b) fertilisation
(c) implantation
(d) none of these.
Answer:
(a) ova formation
Question 21.
What is true about “Saheli” ?
(i) Developed at the CDRI, Lucknow
(ii) Contains a steroidal preparation
(iii) “Once-a-weak” pill
(iv) Many side effects
(v) High contraceptive value
(vi) Very few side effects value
(vii) Low contraceptive value
(a) (i), (ii), (iii), (v), (vi)
(b) (i), (iii), (v), (vi), (vii)
(c) (i), (ii), (iii), (iv), (v)
(d) (i), (iii), (iv), (v)
Answer:
(d) (i), (iii), (iv), (v)
Question 22.
What is the figure given below showing in particular ?

(a) Ovarian cancer
(b) Uterine cancer
(c) Tubectomy
(d) Vasectomy
Answer:
(c) Tubectomy
![]()
Question 23.
Which of the following birth control measures can be considered as the safest ?
(a) The rhythm method
(b) The use of physical barriers
(c) Termination of unwanted pregnancy
(d) Sterilisation techniques
Answer:
(d) Sterilisation techniques
Question 24.
Which of the following is the most widely accepted method of contraception in India at present ?
(a) Cervical caps
(b) Tubectomy
(c) Diaphragms
(d) IUDs (Intra uterine devices)
Answer:
(d) IUDs (Intra uterine devices)
Question 25.
Which of the following is a full proof method contraception ?
(a) Implantation
(b) Lactational amenorrhea
(c) Condoms
(d) Sterilisation
Answer:
(d) Sterilisation
Question 26.
Which of the following statements is correct regarding vasectomy ?
(a) It prevents the production of sperm in the testes.
(b) It prevents the production of semen.
(c) It prevents the movement of sperm into the urethra.
(d) It prevents a man from having an erection.
Answer:
(c) It prevents the movement of sperm into the urethra.
Question 27.
Which of the following statements is incorrect regarding the medical termination of pregnancy (MTP) ?
(a) These help in getting rid of unwanted pregnancies.
(b) These help in aborting the pregnancies which may be harmful to either mother of foetus or both.
(c) These contribute in decreasing the human population.
(d) None of these.
Answer:
(d) None of these.
![]()
Question 28.
Which of the following represents the correct match of a sexually transmitted disease with its pathogen ?
(a) Syphilis-Treponema pallidum
(b) Gonorrhoea-Entamoeba histolytica
(c) Urethritis-Bacillus anthracis
(d) Softsore-Bacillus brevis
Answer:
(a) Syphilis-Treponema pallidum
Question 29.
The sexually transmitted disease, that can affect both the male and the female genitals and may damage the eyes of babies born of infected mothers is
(a) AIDS
(b) syphilis
(c) gonorrhoea
(d) hepatitis
Answer:
(c) gonorrhoea
Question 30.
A sexually transmitted disease symptomised by the development of chancre on the genitals is caused by the infection of
(a) Treponema pallidum
(b) Neisseria gonorrhoeae
(c) human immunodeficiency virus
(d) hepatitis B virus
Answer:
(a) Treponema pallidum
Question 31.
Which of the following is correct regarding HIV, hepatitis B, gonorrhoea, trichomoniasis ?
(a) Trichomoniasis is an STD whereas others are not.
(b) Gonorrhoea is a viral disease whereas others are bacterial.
(c) HIV is a pathogen whereas others are diseases.
(d) Hepatitis B is eradicated completely whereas others are not.
Answer:
(c) HIV is a pathogen whereas others are diseases.
Question 32.
Which one of the following groups includes sexually transmitted diseases caused by bacteria only ?
(a) Syphilis, gonorrhoea, chancroid
(b) Syphilis, chlamydiasis, chancroid
(c) Syphilis, gonorrhoea, scabies
(d) Syphilis, scabies, pediculosis
Answer:
(a) Syphilis, gonorrhoea, chancroid
![]()
Question 33.
Confirmatory test for STDs is
(a) ELISA
(b) PCR
(c) DNA hybridisation
(d) all of these.
Answer:
(d) all of these.
Question 34.
Hepatitis B is transmitted through
(a) blood transfusion
(b) intimate physical contact
(c) sexual contact
(d) all of these
Answer:
(d) all of these
Question 35.
The common means of transmission of AIDS is
(a) sexual intercourse
(b) blood transfusion
(c) placental transfer
(d) all of these.
Answer:
(d) all of these.
Question 36.
Which of the following STDs are not completely curable ?
(a) Chlamydiasis, gonorrhoea, trichomoniasis
(b) Chancroid syphilis, genital warts
(c) AIDS, syphilis, hepatitis B
(d) AIDS, genital herpes, hepatitis B
Answer:
(d) AIDS, genital herpes, hepatitis B
![]()
Question 37.
World AIDS day is
(a) December 21
(b) December 1
(c) November 1
(d) June 11
Answer:
(b) December 1
Question 38.
In vitro fertilisation is a technique that involves transfer of which one of the following into the fallopian tube ?
(a) Embryo only, upto 8 cell stage
(b) Either zygote or early embryo upto 8 cell stage
(c) Embro of 32 cell stage
(d) Zygote only
Answer:
(b) Either zygote or early embryo upto 8 cell stage
Question 39.
The test-tube baby programme employs which one of the following techniques ?
(a) Zygote intra Fallopian transfer (ZIFT)
(b) Intra uterine insemination (IUI)
(c) Gamete intra Fallopian transfer
(d) All of these
Answer:
(a) Zygote intra Fallopian transfer (ZIFT)
Question 40.
The first case of IVF-ET technique success, was reported by
(a) Louis joy Brown and Banting Best
(b) Patrick Steptoe and Rebert Edwards
(c) Robert Steptoe and Gilbert Brown
(d) Baylis and Starling Taylor.
Answer:
(b) Patrick Steptoe and Rebert Edwards
Question 41.
Which of the following is ART ?
(a) IUDs
(b) GIFT
(c) ZIFT
(d) Both (b) & (c)
Answer:
(d) Both (b) & (c)
![]()
Question 42.
The technique called Gamete Intra Fallopian Transfer (GIFT) is recommended for those females
(a) who cannot produce an ovum
(b) who cannot retain the foetus inside uterus
(c) who cannot provide suitable environment for fertilisation
(d) all of these
Answer:
(a) who cannot produce an ovum
Question 43.
Which method can be used for women that cannot produce ovum but can provide suitable environment ?
(a) IUD
(b) GIFT
(c) IUI
(d) ICSI
Answer:
(b) GIFT
Question 44.
Which of these can be used to cure infertility in couples where male partner has very low sperm count ?
(a) IUD
(b) GIFT
(c) IUI
(d) None of these
Answer:
(c) IUI
Question 45.
The method of directly injecting a sperm into ovum in assisted reproductive technology is called
(a) GIFT
(b) ZIFT
(c) ICSI
(d) ET
Answer:
(c) ICSI
Question 46.
Increased IMR and decreased MMR in a population will
(a) cause rapid increase in growth rate
(b) result in decline in growth rate
(c) not cause significant change in growth rate
(d) result in an explosive population.
Answer:
(b) result in decline in growth rate
![]()
Question 47.
Intensely lactating mothers do not generally conceive due to the
(a) suppression of gonadotropins
(b) hypersecretion of gonadotropins .
(c) suppression of gametic transport
(d) suppression of fertilisation.
Answer:
(a) suppression of gonadotropins
Question 48.
A national level approach to build up a reproductively healthy society was taken up in our country in
(a) 1950s
(b) 1960s
(c) 1980s
(d) 1990s
Answer:
(a) 1950s
Question 49.
Emergency contraceptives are effective if used within,
(a) 72 hrs of coitus
(b) 72 hrs of ovulation
(c) 72 hrs of menstruation
(d) 72 hrs of implantation.
Answer:
(a) 72 hrs of coitus
Question 50.
From the sexually transmitted diseases mentioned below, identify the one which does not specifically affect the sex organs.
(a) Syphilis
(b) AIDS
(c) Gonorrhea
(d) Genital warts
Answer:
(b) AIDS
Question 51.
Condoms are one of the most popular contraceptives because of the following reasons.
(a) These are effective barriers for insemination.
(b) They do not interfere with coital act.
(c) These help in reducing the risk of STDs.
(d) All of the above
Answer:
(d) All of the above
Question 52.
The correct surgical procedure as a contraceptive method is
(a) ovariectomy
(b) hysterectomy
(c) vasectomy
(d) castration.
Answer:
(c) vasectomy
![]()